Lexaria Bioscience Finds its Niche in the Lucrative GLP-1 Market

The GLP-1 drug market has quickly become one of the fastest growing and most strategically important sectors in all of pharma. Used mainly to treat Type 2 diabetes and to help manage obesity through weight loss, and dominated by industry leaders like Novo Nordisk and Eli Lilly, the GLP-1 drug class has been propelled into the mainstream by well-known brands like Ozempic® and Wegovy®.
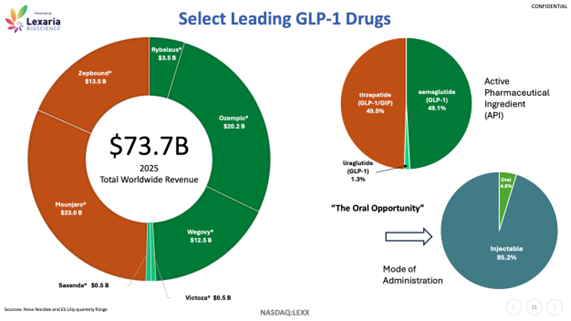
The current market for these drugs is now valued at more than US$70 billion worldwide, and analysts project that figure could climb beyond US$150 - 200 billion by the early 2030s, positioning GLP-1s as one of the largest and most impactful drug classes in pharmaceutical history.
Despite the momentum, key challenges remain. First is tolerability; many patients experience side effects like nausea, vomiting, and gastrointestinal discomfort, which can limit adherence. 50-60% of patients on GLP-1 drugs eventually drop off due to these adverse effects. The second challenge is delivery. Most GLP-1 therapies require patients to self-inject on a regular basis, which can be daunting, is invasive, and can be accompanied by a myriad of unwanted side effects. As a result, the industry is racing to find oral alternatives – aiming to deliver drugs with similar or greater efficacy, and reduced side effects, but in convenient pill form.
One company looking to address both challenges is Lexaria Bioscience (NASDAQ:LEXX), by carving out a unique role within the GLP-1 landscape. Currently in the clinical stage, the company’s proprietary drug delivery platform – DehydraTECHTM – is designed to enable oral dosing and improve tolerability of existing GLP-1 therapies.
DehydraTECH works by pairing the drug with long chain fatty acids and patented processing technology, which help it pass more efficiently through the gut and into the bloodstream, potentially improving absorption and reducing side effects.
While GLP-1 is the company’s current focus, CEO Richard Christopher says there is a broader opportunity for DehydraTECH beyond a single drug class. “It's platform technology, so it's not specifically limited to GLP-1. We've concentrated our resources there because we believe it's our best shot on goal, but the applicability could be much broader, to almost any oral or injectable drug in the world.”
Lexaria Bioscience is advancing a multi-faceted strategy aimed at driving long-term growth. The lead prong of the strategy is a focus on human clinical studies using DehyraTECH in combination with existing active pharmaceutical ingredients (APIs). The objective is to generate compelling data that could attract the attention of major API owners in the GLP-1 space – such as Novo Nordisk, and Eli Lilly – and ultimately support a major licencing or partnership opportunity.
Lexaria already has a Material Transfer Agreement in place with an un-disclosed pharmaceutical partner.
The company has completed five human studies to date, evaluating DehydraTECH with leading GLP-1 compounds like semaglutide (marketed as Ozempic® and Wegovy®, tirzepatide (Mounjaro® and Zepbound®), and liraglutide (Saxenda®). Company reported results have demonstrated consistent reductions in adverse gastrointestinal side effects (see table below).

Beyond partnering with existing drugs, the company is also in development of their own API internally. While their DehydraTECH is designed to work with a wide range of APIs, the company has been developing its own proprietary API in the CBD space, with a focus on hypertension, and has already conducted significant research and early-stage development.
The company is also exploring opportunities with the injectable GLP-1 compound: liraglutide, which lost its patent protection in 2024. Lexaria has conducted DehydraTECH-based evaluations on the drug and sees potential to develop an oral version, which could open the door to a generic oral format.
One of the key industry tailwinds supporting Lexaria is a growing shift in focus within the GLP-1 space – from weight loss efficacy, towards tolerability and long-term drug adherence. CEO Richard Christopher sees this as a huge opportunity. “The differentiating factor now is how do you improve the safety profile, so patients stay on their therapy,” he says. “Even leading drug developers have struggled to fully solve this. That creates a tremendous opportunity for us, given the safety profile DehydraTECH has demonstrated.”
Lexaria’s pursuit of an oral delivery alternative adds another layer to its long-term potential. Today’s GLP-1 market—valued in the tens of billions globally—is still overwhelmingly dominated by injectable therapies, with oral options representing only a small fraction of total worldwide revenues (see chart below). To date, the most notable oral GLP-1 has been Rybelsus from Novo Nordisk, approved for type 2 diabetes. That leaves a significant opportunity for new oral formulations to make a major impact, as patient preference leans heavily towards pills over injections.
While Lexaria can be considered an industry unicorn – a pure play delivery technology company - Halozyme Therapeutics (NASDAQ:HALO) offers the closest comparable business model. They own their own drug delivery platform and partners with major pharma companies, but they are not focused on the lucrative and rapidly growing GLP-1 space.
The dominant players in GLP-1, including Novo Nordisk (NYSE:NVO) and Eli Lilly (NYSE:LLY) are investing heavily in developing oral delivery GLP-1s, but not the delivery mechanism itself. This distinction is what sets Lexaria apart; it’s positioned as a potential add-on technology across multiple existing therapies, rather than a direct competitor to any single drug.
CEO Richard Christopher believes that this positioning could prove highly valuable. “We believe we have game-changing technology on a couple of different fronts. We’re in the sweet spot for a solution for a major challenge in the industry. We feel we have a home run here and we are pushing as hard as we can to find the right partners.”
In a market dominated by major drug developers, Lexaria is positioning itself on a different part of the value chain, and the long-term implications could be significant.
For more information on Lexaria Bioscience Corp. (NASDAQ: LEXX) please click on the request investor info button.
FULL DISCLOSURE: Lexaria Bioscience is a client of BTV-Business Television. This article does not constitute investment advice. Each reader is encouraged to consult with his or her individual financial professional. Any action taken as a result of reading information here is the reader’s sole responsibility.
Latest Posts
Hot Companies
You might also like

Harvest ETFs Launches Two Funds Designed to Thrive in Volatile Times
Investors looking to learn more about the Harvest Premium Yield Canadian Bank ETF or the Harvest Premium Yield Enhanced ETF can go to Harvest ETFs | Equity Income ETFs | Harvest Portfolios Group where you’ll find product information, insight blogs, and videos.





